Ammonium tetrathiomolybdate(TTM,AbMole, M54856)作为一种多功能无机钼硫化合物,在基础科研中展现出多重应用。Ammonium tetrathiomolybdate能螯合铜离子[1],干扰铜依赖性通路(如抑制ATP7B铜转运蛋白表达、降低赖氨酰氧化酶活性及胶原交联),并抑制铜死亡;Ammonium tetrathiomolybdate(TTM,CAS No.:15060-55-6)同时也能作为硫化氢供体,通过抑制线粒体氧化磷酸化、减少活性氧生成发挥调控作用[2]。此外,Ammonium tetrathiomolybdat(TTM,AbMole, M54856)e还被证实可激活NRF2通路,抑制铜死亡相关效应分子、并调控Akt/glucose摄取与乳酸代谢通路[1, 3]。
在细胞模型研究中,Ammonium tetrathiomolybdate(四硫代钼酸盐)在1.5 μM浓度下处理犬血管肉瘤细胞系DEN-HSA,表现出显著的细胞毒性[4]。Ammonium tetrathiomolybdate在HepG2人肝癌细胞与HEK293人胚肾细胞中,以1 μM–10 mM的浓度梯度处理24小时可观察到细胞特异性铜保留差异及氧化应激反应[5]。动物实验方面,Ammonium tetrathiomolybdate(TTM)在C57BL/6小鼠胆管结扎纤维化模型中,每日0.9 mg/kg剂量干预可缓解纤维化进程[6],此外,在野百合碱(Monocrotaline)诱导的肺纤维化绵羊模型中,Ammonium tetrathiomolybdate(TTM)干预可抑制血管生成与纤维化[7]。在TNBC(三阴性乳腺癌)小鼠模型中,口服Ammonium tetrathiomolybdate(TTM)能维持血清铜蓝蛋白于8–17 mg/dL的水平,显著抑制肺转移、降低LOX活性及胶原交联[8]。
范例详解
Front Oncol. 2025 Oct 10;15:1532772.
AbMoel的Ammonium tetrathiomolybdate(TTM,AbMole, M54856)在本研究中作为铜死亡的抑制剂,通过对照实验以验证 FOXJ1 基因对宫颈癌细胞铜死亡的调控作用。研究结果表明,FOXJ1基因过表达显著抑制宫颈癌细胞增殖、侵袭和迁移,并促进铜死亡,而TTM通过螯合铜离子,抑制了铜死亡过程,从而逆转了FOXJ1过表达的肿瘤抑制作用。
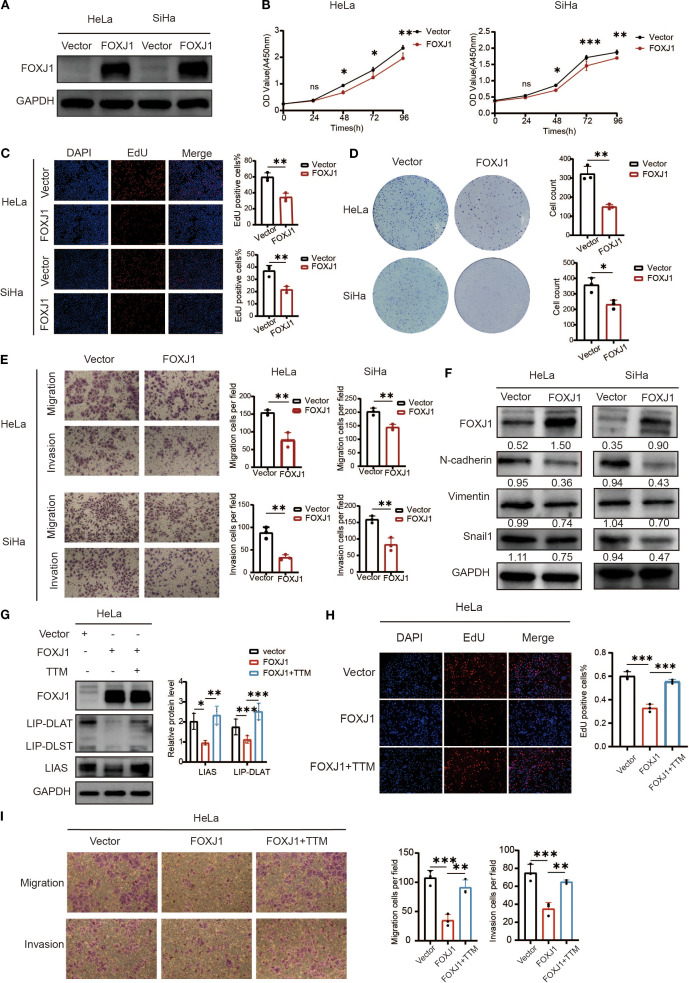
图1. Overexpression of FOXJ1 decreased the proliferation, invasion, migration and the level of EMT process through the regulation of cuproptosis[9]
参考文献及鸣谢
[1] Ryumon, S.; Okui, T.; Kunisada, Y.; et al. Ammonium tetrathiomolybdate enhances the antitumor effect of cisplatin via the suppression of ATPase copper transporting beta in head and neck squamous cell carcinoma. Oncology reports 2019, 42 (6), 2611-2621.
[2] Durham, T.; Zander, D.; Stomeo, N.; et al. Chemistry, pharmacology, and cellular uptake mechanisms of thiometallate sulfide donors. British journal of pharmacology 2020, 177 (4), 745-756.
[3] Navratilova, J.; Karasova, M.; Kohutkova Lanova, M.; et al. Selective elimination of neuroblastoma cells by synergistic effect of Akt kinase inhibitor and tetrathiomolybdate. Journal of cellular and molecular medicine 2017, 21 (9), 1859-1869.
[4] Sloan, C. Q.; Rodriguez, C. O., Jr. In vitro effects of doxorubicin and tetrathiomolybdate on canine hemangiosarcoma cells. American journal of veterinary research 2018, 79 (2), 219-225.
[5] Sachdeva, S.; Maret, W. Comparative outcomes of exposing human liver and kidney cell lines to tungstate and molybdate. Toxicology mechanisms and methods 2021, 31 (9), 690-698.
[6] Song, M.; Song, Z.; Barve, S.; et al. Tetrathiomolybdate protects against bile duct ligation-induced cholestatic liver injury and fibrosis. The Journal of pharmacology and experimental therapeutics 2008, 325 (2), 409-416.
[7] Derseh, H. B.; Perera, K. U. E.; Dewage, S. N. V.; et al. Tetrathiomolybdate Treatment Attenuates Bleomycin-Induced Angiogenesis and Lung Pathology in a Sheep Model of Pulmonary Fibrosis. Frontiers in pharmacology 2021, 12, 700902.
[8] Chan, N.; Willis, A.; Kornhauser, N.; et al. Influencing the Tumor Microenvironment: A Phase II Study of Copper Depletion Using Tetrathiomolybdate in Patients with Breast Cancer at High Risk for Recurrence and in Preclinical Models of Lung Metastases. Clinical cancer research : an official journal of the American Association for Cancer Research 2017, 23 (3), 666-676.
[9] Cui, Y.; Liu, Z.; Zhang, L.; et al. Construction of a novel cuproptosis-related gene signature for predicting microenvironment, prognosis and therapeutic response in cervical cancer. Frontiers in oncology 2025, 15, 1532772.
细胞实验参考
细胞系:AML cell cervical cancer cells
方法: The pENTER vector was used as a negative control. Ammonium tetrathiomolybdate (TTM) was purchased from AbMole (CAS No.:15060-55-6). Cells were treated with TTM at a final concentration of 40μM for 2 hours.
浓度:40μM
处理时间:2 h.
参考文献:Front Oncol. 2025 Oct 10;15:1532772.
* 上述方法来自公开文献,仅供相同目的实验参考。如实验目的、材料、方法不同,请参考其他文献。
动物实验参考
动物模型:C57BL/6N mice
配制:Distilled water
剂量:30 mg/kg
给药处理:Oral gavage
参考文献:Am J Physiol Heart Circ Physiol. 2011 Sep;301(3):H712-20.
* 上述方法来自公开文献,仅供相同目的实验参考。如实验目的、材料、方法不同,请参考其他文献。
体内实验的工作液,建议现用现配,当天使用;如在配制过程中出现沉淀、析出现象,可以通过超声和(或)加热的方式助溶。切勿一次性将产品全部溶解。
建议制定动物给药及实验方案时,尽量参考已发表的相关实验文献(溶剂种类及配比众多,简单地溶解目的化合物,并不能解决动物给药依从性、体内生物利用度、组织分布等相关问题,未必能保证目的化合物在动物体内充分发挥生物学效用)。
转载本文请联系原作者获取授权,同时请注明本文来自王开来科学网博客。
链接地址:https://wap.sciencenet.cn/blog-3646309-1525185.html?mobile=1
收藏

