博文
研究人员报告了颠覆镁化学的发现
||
研究人员报告了颠覆镁化学的发现
诸平
据克里斯·巴顿(Chris Button)2021年4月29日提供的消息,德国埃尔朗根-纽伦堡大学(全名弗里德里希-亚历山大 埃尔朗根-纽伦堡大学,德文:Friedrich-Alexander-Universität Erlangen-Nürnberg或者英文:Friedrich-Alexander University Erlangen-Nürnberg)化学家,于2021年4月28日在《自然》(Nature)杂志网站上发表了镁化学方面的突破性研究成果——B. Rösch, T. X. Gentner, J. Eyselein, J. Langer, H. Elsen, S. Harder. Strongly reducing magnesium(0) complexes. Nature, Published: 28 April 2021. Volume 592, pages: 717–721. DOI: 10.1038/s41586-021-03401-w. https://www.nature.com/articles/s41586-021-03401-w
镁(Mg)是地球上富含的一种早期主族金属元素,具有低电负性,因此容易失去价电子,与其它元素结合使用时,它自然仅以最稳定的形式作为带正电的Mg2+阳离子出现。Mg2+阳离子存在于各种矿物质中,但在叶绿素中也存在。首次在星际云(interstellar cloud)中检测到异常的镁氧化态(Mg+),但最近发现了第一个与Mg+形成的配合物,并被隔离。
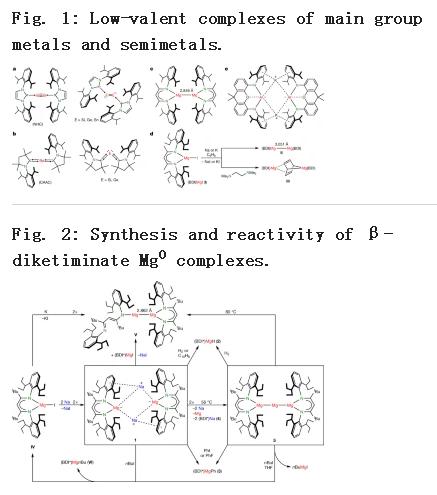
无机和有机金属化学首席教授Sjoerd Harder领导的团队报告首次发现了镁零价态(Mg0)。有一种配合物其中镁的氧化态就是零价(Mg0),且整个配合物带负电荷。这些配合物包含独特的镁-钠键(Mg-Na),其反应与普通Mg2+化合物完全不同。电子不足的Mg2+阳离子可以接受电子,而富含电子的阴离子中的Mg0通过提供电子参与反应。
该Mg0配合物可溶于常见的有机溶剂,并且是一种非常强的还原剂。稍加加热即可立即还原Na+阳离子使其形成Na0。众所周知,钠虽然是一种金属,但极易被氧化成Na+阳离子。在热分解期间,形成了一种新型的配合物,其中三个Mg原子像珠链一样连接。此Mg3团簇的反应类似于原子Mg0,可以看作是Mg金属的最小部分,但是它可溶于有机溶剂。这类新型的阴离子Mg配合物完全颠倒了Mg的化学性质。可以预期这种可溶的,高效还原剂的进一步异常反应性。上述介绍仅供参考,欲了解更多信息敬请注意浏览原文或者相关报道。
化学家创造了新的氧化剂作为制备化学的工具(Chemists create new oxidizers as a tool for preparative chemistry)
A complex of a metal in its zero oxidation state can be considered a stabilized, but highly reactive, form of a single metal atom. Such complexes are common for the more noble transition metals. Although rare examples are known for electronegative late-main-group p-block metals or semimetals1,2,3,4,5,6, it is a challenge to isolate early-main-group s-block metals in their zero oxidation state7,8,9,10,11. This is directly related to their very low electronegativity and strong tendency to oxidize. Here we present examples of zero-oxidation-state magnesium (that is, magnesium(0)) complexes that are stabilized by superbulky, monoanionic, β-diketiminate ligands. Whereas the reactivity of an organomagnesium compound is typically defined by the nucleophilicity of its organic groups and the electrophilicity of Mg2+ cations, the Mg0 complexes reported here feature electron-rich Mg centres that are nucleophilic and strongly reducing. The latter property is exemplified by the ability to reduce Na+ to Na0. We also present a complex with a linear Mg3 core that formally could be described as a MgI–Mg0–MgI unit. Such multinuclear mixed-valence Mgn clusters are discussed as fleeting intermediates during the early stages of Grignard reagent formation. Their remarkably strong reducing power implies a rich reactivity and application as specialized reducing agents.
https://wap.sciencenet.cn/blog-212210-1284631.html
上一篇:肠道真菌:新型化学品的意外来源
下一篇:双孢菇粉片可否减慢前列腺癌的进展?