博文
植物多胺(Plant Polyamines)(二)
|
植物中的多胺代谢
1.植物中多胺的合成代谢
植物中,腐胺(Put)合成的第一步限速反应是由精氨酸脱羧酶(arginine decarboxylase, ADC)催化精氨酸,或者是由鸟氨酸脱羧酶(ornithine decarboxylase, ODC)催化鸟氨酸的脱羧反应。关于拟南芥中是否存在鸟氨酸途径,依旧存有争议。不少人在拟南芥中尝试克隆鸟氨酸脱羧酶基因(ODC)但都失败了(Hanfrey et al., 2001),然而,Tasssoni等人(2003)却在拟南芥中检测到了假定鸟氨酸脱羧酶的活性。亚精胺合成酶(SPDS, EC 2.5.1.16)催化了从腐胺到亚精胺的反应,将氨丙基转移到胺受体上形成亚精胺,而后,再由精胺合成酶(SPMS, EC 2.1.22)进一步催化合成了精胺(Alcázar et al., 2010)。氨丙基基团的另一个供体是脱羧S-腺苷甲硫氨酸(decarboxylated S-adenosylmethionine, dcSAM),S-腺苷甲硫氨酸(S-adenosylmethionine, SAM)由SAM脱羧酶催化(SAM decarboxylase, SAMDC; EC 4.1.1.50)得到dcSAM(Slocum et al., 1984)。(见图2)
拟南芥中,多胺合成酶都是由多个基因编码。例如,编码精氨酸脱羧酶ADC的有两个基因,分别是ADC1和 ADC2,编码亚精胺合成酶的有两个,SPDS1和SPDS2,编码精胺合成酶SPMS的也有两个,SPMS和acaulis 5 (ACL5)。SAMDC的编码基因则有四个,SAMDC1 到SAMDC4(见图2)(Takahashi and Kakehi, 2010)。
2.植物中多胺的分解代谢
多胺的分解涉及到一个或者多个二胺氧化酶(diamine oxidases, DAO, EC 1.4.3.6)和(含黄素腺嘌呤二核苷酸)多胺氧化酶[(flavin adenine dinucleotide)FAD-containing polyamine oxidases, PAO; EC 1.5.3.11)]。这些氧化酶将碳氧化后连接到次级或者初级氨基团上(Moschou et al., 2012; Tavladoraki et al., 2012; 2016)。拟南芥含有五个多胺氧化酶基因,分别是AtPAO1 到 AtPAO5,编码位于细胞质或者过氧化物酶体上的多胺氧化酶 (Tavladoraki et al., 2006; Kamada-Nobusada et al., 2008; Takahashi and Kakehi, 2010; D. W. Kim et al., 2014)。包括拟南芥在内的一些物种,精胺由PAO氧化为亚精胺,而后再氧化为腐胺(Put),并产生过氧化氢(H2O2)和3-氨基丙醛(3-aminopropanal)(Cona et al., 2006)。多胺的这种合成与氧化分解双向代谢途径在植物比较常见(见图2)(Moschou et al., 2008)。
二胺氧化酶(DAO),是位于质外体或者过氧化物酶体上的含有铜的氨氧化酶(CuAOs)。通常可将二氨腐胺或者尸胺氧化分解为初级氨基基团,产生Δ1- 吡咯啉(Δ1-pyrroline)(后进入三羧酸循环),过氧化氢(H2O2)和氨离子(NH4+)(Cohen, 1998; Alcázar et al., 2010)。拟南芥含有10个CuAOs编码基因[AtCuAOα1, AtCuAOα2, AtCuAOα3 (AtCuAO2), AtCuAOβ (ATAO1 or AtAO1), AtCuAOγ1 (AtCuAO1), AtCuAOγ2, AtCuAOδ (AtCuAOδ2), AtCuAOζ (AtCuAO3), AtCuAOε1 (AtCuAOε) and AtCuAOε2 (AtCuAOδ1)](Planas-Portell et al., 2013; Ghuge et al., 2015; Tavladoraki et al., 2016)。
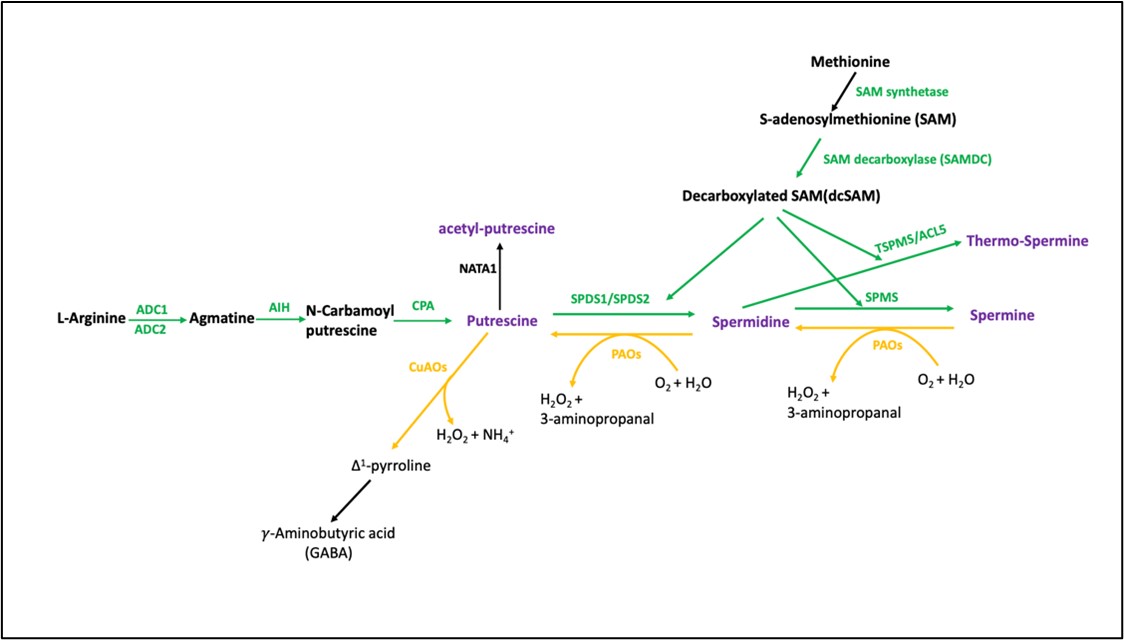
图2. 植物中多胺代谢 (Alcázar et al., 2010)。ADC, arginine decarboxylase; AIH, agmatine iminohydrolase; CPA, N-carbamoylputrescine amidohydrolase; NATA1: N-Acetyltransferase activity 1; SPDS, spermidine synthase SPMS, spermine synthase; ACL5, acaulis5; TSPMS, thermo -spermine synthase; SAMDC, S-adenosylmethionine decarboxylase; PAO , polyamine oxidases; CuAO, copper-containing amine oxidases.
参考文献
Alcázar R, Altabella T, Marco F, Bortolotti C, Reymond M, Koncz C, Carrasco P, Tiburcio AF (2010) Polyamines: Molecules with regulatory functions in plant abiotic stress tolerance. Planta 231: 1237–1249
Cona A, Rea G, Angelini R, Federico R, Tavladoraki P (2006) Functions of amine oxidases in plant development and defence. Trends Plant Sci 11: 80–8
Ghuge SA, Carucci A, Rodrigues-Pousada RA, Tisi A, Franchi S, Tavladoraki P, Angelini R, Cona A (2015) The apoplastic copper amine oxidase1 mediates jasmonic acid-induced protoxylem differentiation in Arabidopsis roots. Plant Physiol 168: 690–707
Hanfrey C, Sommer S, Mayer MJ, Burtin D, Michael AJ (2001) Arabidopsis polyamine biosynthesis: Absence of ornithine decarboxylase and the mechanism of arginine decarboxylase activity. Plant J 27: 551–560
Kamada-Nobusada T, Hayashi M, Fukazawa M, Sakakibara H, Nishimura M (2008) A putative peroxisomal polyamine oxidase, AtPAO4, is involved in polyamine catabolism in Arabidopsis thaliana. Plant Cell Physiol 49: 1272–82
Kim DW, Watanabe K, Murayama C, Izawa S, Niitsu M, Michael AJ, Berberich T, Kusano T (2014) Polyamine oxidase5 regulates Arabidopsis growth through thermospermine oxidase activity. Plant Physiol 165: 1575–1590
Moschou PN, Sanmartin M, Andriopoulou AH, Rojo E, Sanchez-Serrano JJ, Roubelakis-Angelakis KA (2008) Bridging the gap between plant and mammalian polyamine catabolism: A novel peroxisomal polyamine oxidase responsible for a full back-conversion pathway in Arabidopsis. Plant Physiol 147: 1845–1857
Moschou PN, Wu J, Cona A, Tavladoraki P, Angelini R, Roubelakis-Angelakis KA (2012) The polyamines and their catabolic products are significant players in the turnover of nitrogenous molecules in plants. J Exp Bot 63: 5003–5015
Planas-Portell J, Gallart M, Tiburcio AF, Altabella T (2013) Copper-containing amine oxidases contribute to terminal polyamine oxidation in peroxisomes and apoplast of Arabidopsis thaliana. BMC Plant Biol 13: 109
Seiler N (1999) A Guide to the Polyamines . Seymour S. Cohen. Q Rev Biol 74: 342–343
Slocum RD, Kaur-Sawhney R, Galston AW (1984) The physiology and biochemistry of polyamines in plants. Arch Biochem Biophys 235: 283–303
Takahashi T, Kakehi JI (2010) Polyamines: Ubiquitous polycations with unique roles in growth and stress responses. Ann Bot 105: 1–6
Tassoni A, Fornalè S, Bagni N (2003) Putative ornithine decarboxylase activity in Arabidopsis thaliana: Inhibition and intracellular localisation. Plant Physiol Biochem 41: 871–875
Tavladoraki P, Cona A, Angelini R (2016) Copper-containing amine oxidases and FAD-dependent polyamine oxidases are key players in plant tissue differentiation and organ development. Front Plant Sci 7: 824
Tavladoraki P, Cona A, Federico R, Tempera G, Viceconte N, Saccoccio S, Battaglia V, Toninello A, Agostinelli E (2012) Polyamine catabolism: Target for antiproliferative therapies in animals and stress tolerance strategies in plants. Amino Acids 42: 411–426
Tavladoraki P, Rossi MN, Saccuti G, Perez-Amador MA, Polticelli F, Angelini R, Federico R (2006) Heterologous expression and biochemical characterization of a polyamine oxidase from Arabidopsis involved in polyamine back conversion. Plant Physiol 141: 1519–1532
[RAH1]Thermospermine is derived from Spd not Spm.
[RAH2]In Arabidopsis, PAOs do not catalyze terminal catabolism (shown in figure) but back-conversion reactions.
https://wap.sciencenet.cn/blog-3464502-1278780.html
上一篇:植物多胺(Plant Polyamines)(一)